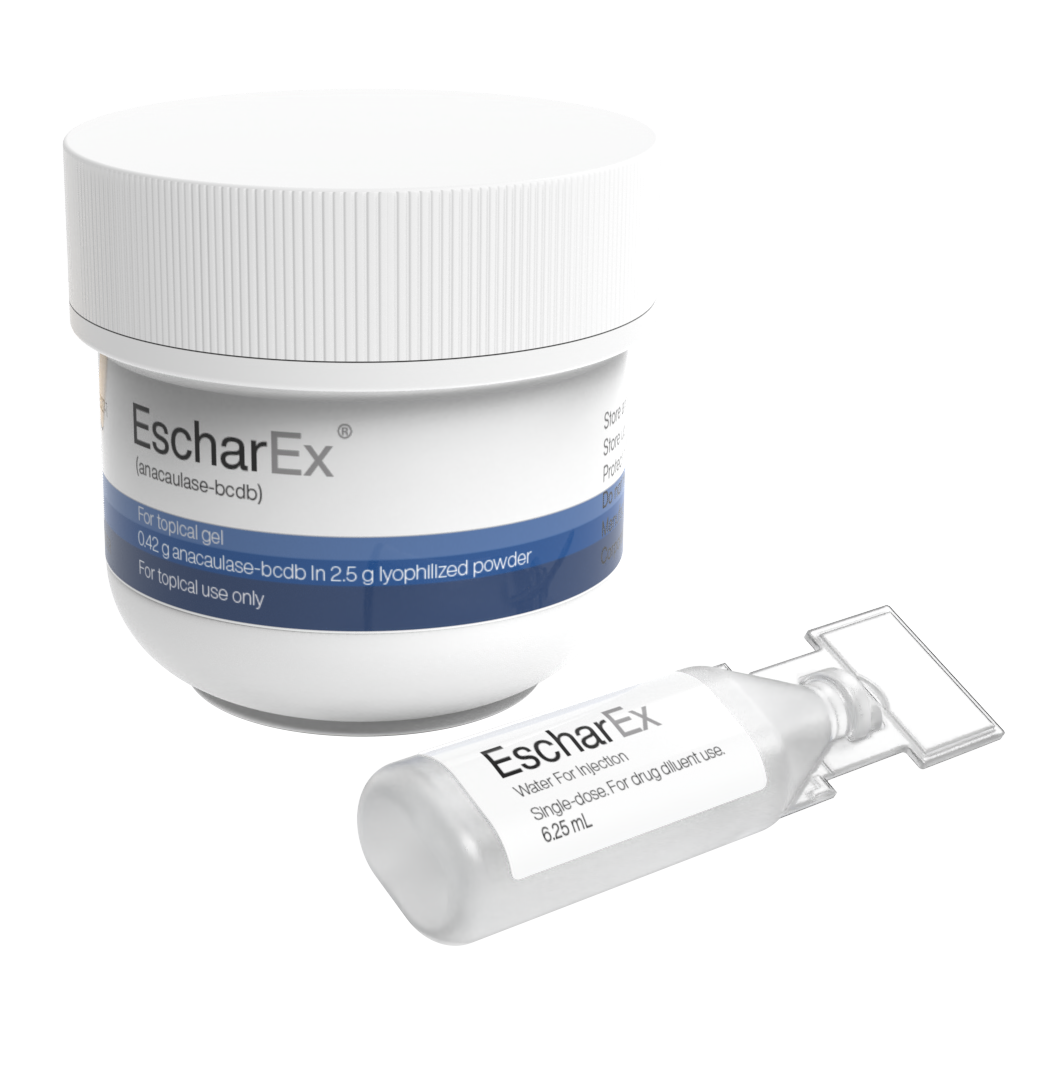
EscharEx® is a bromelain-based, bioactive enzymatic therapy in advanced clinical development for the debridement of chronic and hard-to-heal wounds. Designed for topical, once-daily application, EscharEx has demonstrated a favorable safety profile and effective wound bed preparation in multiple Phase II trials. The therapy has shown the ability to remove non-viable tissue, promote granulation tissue, and reduce bioburden and biofilm. A global Phase III study in venous leg ulcers (VLUs) is currently underway, and a clinical study in diabetic foot ulcers (DFUs) is in preparation. EscharEx has shown clinical advantages over the leading enzymatic debridement product and targets a substantial global market opportunity.
EscharEx® development is being supported in part by the European Innovation Council (EIC) through its accelerator program for the indication of diabetic foot ulcers.